




Medical CNC Machined Parts
Medical CNC Machined Parts
Medical CNC machined parts are crafted from medical-grade premium materials using state-of-the-art 3-axis, 4-axis, and 5-axis CNC machining centers. From simple surgical instrument components to complex orthopedic implant geometries, we handle custom medical orders with ultra-precision, ensuring each part adheres to your exact specifications, medical safety standards, and biocompatibility requirements.
Key Features
Superior Medical-Grade Precision
Achieve tight tolerances of ±0.001mm, ensuring consistent fit and function across all batches—critical for medical devices where even minor deviations can impact patient safety. Our advanced CNC systems, paired with medical-specific quality control processes, eliminate errors, guaranteeing part uniformity and reliability for critical healthcare applications.
Biocompatible Material Compatibility
Process a full range of medical-grade materials, including titanium (Grade 2, Grade 5), stainless steel (316L, 17-4PH), aluminum (6061 medical grade), PEEK, medical-grade plastics (ABS medical grade, PC medical grade), and biocompatible exotic alloys. We select materials based on your medical application requirements, balancing biocompatibility, corrosion resistance, sterilizability, mechanical strength, and cost-effectiveness.
Complex Geometry Capability
5-axis CNC machining enables us to produce medical parts with intricate shapes, undercuts, and multi-angle features that traditional machining methods cannot achieve—ideal for high-performance surgical instruments, orthopedic implants, and diagnostic equipment components.
High Efficiency & Fast Turnaround
Automated medical CNC processes reduce production time while maintaining strict quality and compliance standards. We offer flexible production runs, from medical prototypes (for device testing and validation) to large-scale mass production, with competitive lead times to meet your medical device development and production deadlines.
Medical-Grade Surface Finish
Optional medical-specific surface treatments (passivation, electropolishing, anodizing, polishing, deburring) ensure a smooth, wear-resistant, and contamination-free surface that enhances biocompatibility, aesthetics, and part lifespan. Standard surface finishes include Ra 0.8-Ra 3.2, with custom finishes available to meet specialized medical requirements.
Technical Specifications
| Machining Type | 3-axis, 4-axis, 5-axis CNC Milling & Turning (Medical-Specific Calibration) |
|---|---|
| Tolerance | ±0.001mm ~ ±0.01mm (Customizable based on medical device requirements) |
| Material Range | Titanium (Grade 2, Grade 5), Stainless Steel (304, 316L, 17-4PH), Aluminum (6061 Medical Grade), Brass (Medical Grade), Copper (Medical Grade), PEEK, Medical-Grade Plastics (ABS, POM, PC, Nylon), Biocompatible Exotic Alloys |
| Maximum Workpiece Size | 500mm × 500mm × 500mm (L×W×H) - Larger sizes available upon medical project request |
| Surface Finish | Ra 0.8-Ra 3.2 (Standard); Passivation, Electropolishing, Anodizing, Plating, Polishing (Medical-Specific Optional Treatments) |
| Production Capacity | Prototype: 1-10 Pieces (for Medical Validation); Small Batch: 10-1000 Pieces; Mass Production: 1000+ Pieces |
| Quality Control | CMM (Coordinate Measuring Machine), Optical Comparator, Hardness Tester, Surface Roughness Tester, Biocompatibility Testing (Optional) |
Applications
Surgical Instruments
Laparoscopic instrument parts, orthopedic surgical tools, neurosurgical components, minimally invasive surgical device parts, scalpel handles, and forceps components
Diagnostic Equipment
MRI machine components, CT scanner precision parts, ultrasound device components, blood analyzer parts, and diagnostic probe housings.
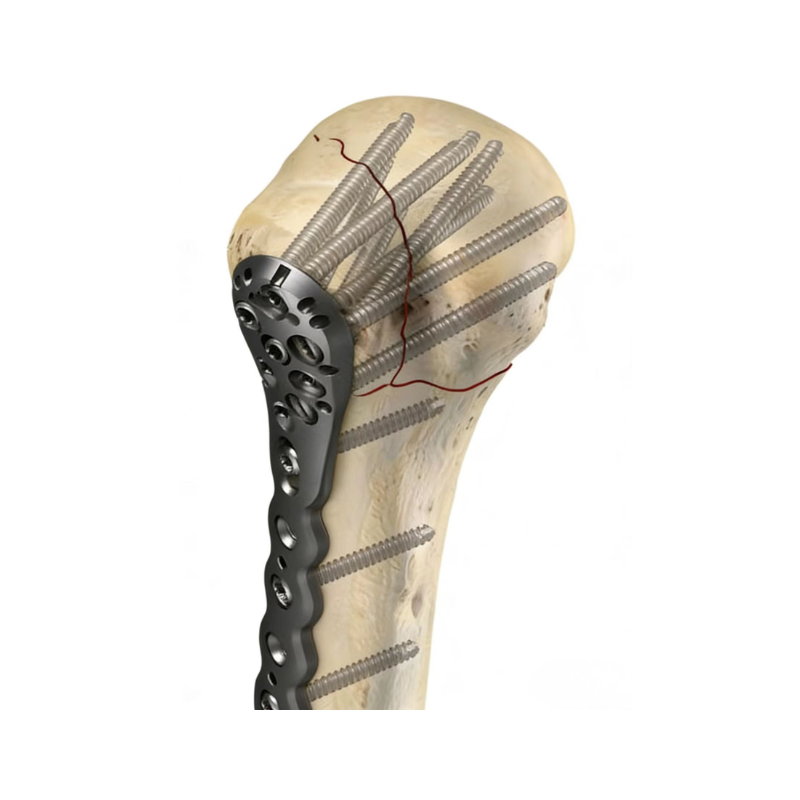
Orthopedic & Implant Devices
Hip/knee/spine implant components, surgical fixation devices, prosthetic parts, and orthopedic bracket components
Patient Care Equipment
Infusion pump parts, ventilator components, patient monitor parts, dialysis machine components, and mobility aid parts
Lab & Research Equipment
Laboratory centrifuge parts, pipette components, sample processor parts, and medical analytical instrument components
Quality Assurance & Compliance
Each part is rigorously tested using precision measuring tools (CMM, optical comparator) to verify dimensions, tolerances, and surface finish. Biocompatibility testing (optional) and full traceability (from raw material to delivery) are maintained to ensure compliance with ISO 13485, FDA, and CE MDR standards. Only qualified parts pass inspection and are delivered.